BD Sodium Chloride 0.9% NACL IV Bag 1000ml, Case (1727170107) *R*
Product Code: 1727170107
The BD 1727170107 Sodium Chloride IV Bag (1000ml) is a sterile, non‑pyrogenic saline solut..
BD Sodium Chloride 0.9% NACL IV Bag 500ml, Case (1727170106) *R*
Product Code: 1727170106
The BD 1727170106 Sodium Chloride IV Bag (500ml) is a sterile, non‑pyrogenic saline solution fo..
BD Sodium Chloride 0.9% NACL IV Bag 250ml, Case (1727170105) *R*
Product Code: 1727170105
The BD 1727170105 Sodium Chloride IV Bag (250ml) is a sterile, non‑pyrogenic saline soluti..
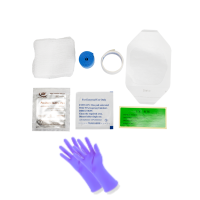
Dynarex IV Start Kit With Pair of Nitrile Gloves, Case (4704)
Product Code: 4704
Dynarex 4704 IV Start Kit – Sterile, Efficient, Ready-to-Use The Dynarex 4704 IV ..
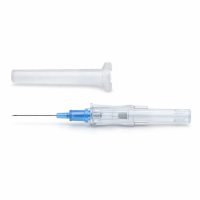
ICU Medical ProtectIV IV Catheter 22 Gauge X 1 *R* (3060)
Product Code: 3060
The ICU Medical 3060 Protectiv® Plus Safety IV Catheter is engineered to reduce accidental ..
Riserva VGPF Medical Grade Vinyl Exam Gloves (Box of 150)
Product Code: VGPF
Riserva Disposable Vinyl Protective Gloves deliver dependable, latex‑free protection designed for hi..
Dukal Alcohol Preps Medium Sterile 2-Ply, Box (853)
Product Code: 853
The Dukal 853 Sterile Alcohol Prep Pad is a professional‑grade antiseptic cleansing pad designed for..
PDI Super Sani-Cloth Germicidal Surface Wipe Purple Top, Tub (Q55172)
Product Code: PDI-Q55172
The PDI Super Sani-Cloth Germicidal Wipe, Tub of 160 (Q55172) is a top-choice disinfecting solution ..
BD PosiFlush Prefilled Normal Saline Flush 10ml, Box (306546) *R*
Product Code: 306546
The BD 306546 PosiFlush™ Prefilled Normal Saline Flush Syringe is engineered to support best‑p..
Nuance CryoDose TA Topical Anesthetic Mist Spray 115ml Can (1802)
Product Code: 1802
The Nuance CryoDose TA Topical Anesthetic Mist Spray (1802) is a fast-acting, non-flammable anesthet..

Midmark Ritter 204 Exam Table with Upholstery Top
Product Code: 204
Midmark 204 Exam Table bases are in stock ready to ship. We offer quick ship for the uphol..
Tuttnauer T-Top Autoclave Sterilizer
Product Code: T-TOP
The Tuttnauer T‑Top Autoclave is a next‑generation Class S tabletop sterilizer engineered for clinic..
Welch Allyn Green Series 777 Integrated Wall System, Configuration 777-PM2WXS-US
Product Code: 777-PM2WXS-US
Configuration 777-PM2WXS-US includes PanOptic Basic LED Ophthalmoscope, MacroView Basic LED Oto..
NDD 2500-2A, EasyOne Air Portable and PC Spirometer
Product Code: 2500-2
The EasyOne Air is a modern, portable spirometry system designed to deliver accurate, reliable lung‑..
Healthometer ADA Compliant Digital Platform Scale with Extra Wide Handrails (1100KL)
Product Code: 1100KL
Health o meter® 1100KL Digital Platform Scale with Extra‑Wide Handrails The Health o meter 11..
Welch Allyn Green Series 600 Minor Procedure Light with Wall Mount (44610)
Product Code: 44610
The Welch Allyn 44610 Green Series 600 Minor Procedure Light is engineered for clinical environments..
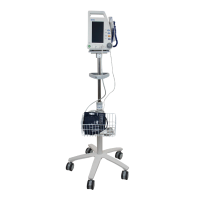
Dynarex Vital Signs Patient Monitor (10841)
Product Code: 10841
The Dynarex 10841 Vital Signs Monitor is a compact, touchscreen‑driven patient monitoring system des..
Welch Allyn VS100 Spot Vision Screener (VS100S-B)
Product Code: VS100S-B
The Hillrom / Welch Allyn Spot Vision Screener VS100S‑B is a handheld, portable, FDA‑cleared vision ..
Midmark Ritter Side Chair (280-001)
Product Code: 280-001
Midmark Ritter 280 Side Chair — Durable, Comfortable Seating for Clinical Spaces The Midmar..
Midmark Digital ECG (4-000-0080)
Product Code: 4-000-0080
Midmark 4‑000‑0080 Digital ECG (DICOM‑Compatible) Current Promotion from March 2, 2026 ..
- Melva G.
 Google Reviews
Google Reviews